Juan Soto may have been traded away from Washington (tear) but the good (?) news for political fans is that the town’s regulatory MVP is staying put.
In a city where every legislative and executive branch office thinks that they are critically important, there is one true superstar: the Office of Information and Regulatory Affairs (OIRA).
While the power source differs from administration to administration, OIRA has withstood the test of time and remained king of the swamp. However, many have never heard of OIRA and its impact on Americans’ everyday lives.
Situated in the Office of Management and Budget (OMB), OIRA oversees the regulatory process for all executive agencies, from reviewing and shaping agency drafts to determining the regulatory actions that can move forward. Think of OIRA as the all-powerful umpire, calling balls, strikes, and outs on the final regulations each department promulgates. OIRA strives to ensure each agency does not expand beyond their true capacity. The president or a cabinet secretary may desire a certain regulatory outcome, but that regulation must run through the very meticulous OIRA process before reaching the bottom of the ninth.
As the country braces for the midterm elections and the possibility of Republicans seizing control of the House of Representatives and possibly the Senate, OIRA’s already sizeable influence could prove even more consequential. Without a Democratic led Congress, President Biden will likely rely on executive actions and the regulatory process to deliver his policy objectives in the runup to the 2024 presidential election. But it’s not as simple as directing cabinet secretaries to deliver with that little understood federal umpire standing in the way.
Here is a who, what and why on OIRA that you need to know.
What is OIRA?
OIRA was established by Congress through the Paperwork Reduction Act of 1980 as an answer to supporters of deregulation who felt that collecting information for federal agencies was a cumbersome burden for businesses, nonprofits, and local governments alike. The Act was passed with the intention of lessening this burden by requiring executive agencies to obtain approval from the newly formed OIRA before attempting to collect information from the public, so that only essential data would be collected.
Since then, the functions of OIRA have expanded dramatically via numerous executive orders (EO) from presidents on both sides of the aisle—not acts of Congress. In 1981, President Reagan issued an EO that required agencies to submit all major regulations, on top of information collection requests, to OIRA for review. This was an action to help the Reagan Administration not only corral government bureaucracy but ensure the regulatory process was data driven, produced intended results, limited unintended consequences, and mirrored their overall government objectives to the best degree possible. Not unexpectedly, this led to significant controversy, as critics expressed concerns that OIRA lacked transparency and elevated the power of the executive branch. President Clinton attempted to alleviate such concerns in 1993 by issuing EO 12866. Although it reaffirmed the need for OIRA to evaluate and approve major regulatory actions, this EO also confirmed the “primacy” of federal agencies and set the goal of improving coordination, legitimacy, transparency, and accessibility within the regulatory process. This landmark EO guides OIRA’s regulatory process today.
What is OIRA’s Regulatory Process Like?
Like a baseball game, the regulatory process under OIRA can be slow and steady but does present opportunities to score runs.
First, OIRA decides which proposed regulatory actions are “significant” and subject to review. Annually, OIRA typically classifies around 500-700 regulatory actions as significant.

Have an annual effect on the economy of $100 million or more or adversely affect in a material way the economy, a sector of the economy, productivity, competition, jobs, the environment, public health or safety, or state, local, or tribal governments or communities;

Create a serious inconsistency or otherwise interfere with an action taken or planned by another agency;

Materially alter the budgetary impact of entitlements, grants, user fees, or loan programs or the rights and obligations of recipients thereof; or

Raise novel legal or policy issues arising out of legal mandates, the president’s priorities, or the principles set forth in this EO.
Once categorized as significant, OIRA oversees its movement throughout the regulatory process, from proposed rule to final rule. There are several hurdles that executive agencies must overcome prior to regulatory actions taking effect. During the draft phase, OIRA requires agencies to conduct cost-benefit analyses of regulatory actions and only allows actions to move forward when the benefits outweigh the costs. OIRA can also require agencies to reconsider regulatory actions in the draft phase for a variety of reasons, including if OIRA finds the cost-benefit analysis to be insufficient.
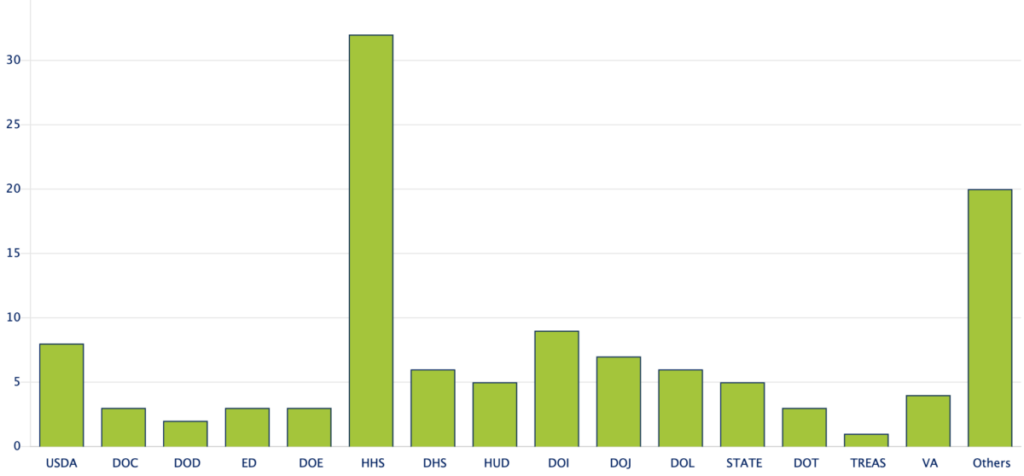
In line with EO 12866, OIRA offers to meet with any party who wishes to discuss specific regulatory actions under review as a mechanism for boosting public access to the regulatory process. In an effort to provide greater transparency, OIRA also maintains a regulatory dashboard with status updates. Currently, there are 117 regulatory actions under review, 32 of which come from the Department of Health and Human Services – the most of any executive agency. OIRA has 90 days to review regulatory actions but can extend this period by up to 30 days.
Regulatory Actions Currently Under Review By Agency
What’s an OIRA Meeting Like?
Take it from someone who has participated in OIRA meetings over the years – it’s the World Series of meetings and complaining to the most powerful umpire in Washington will not produce a favorable outcome.
OIRA meetings are all about facts and data. Interested parties will need to present their views on why the potential regulation should be approved or altered, why it fails to live up to legislative intent, or possibly exceeds agency authority, and why it doesn’t measure up to the cost benefit analysis. OIRA staff may raise concerns with what a department or agency did or did not consider when drafting the regulation.
I have often joked that if an OIRA official asks a question in the meeting, your meeting preparation was on point! One might characterize a successful regulatory meeting as one that elicits questions that result in OIRA asking the department promulgating the regulation if they considered certain impacts, policy options, cost or benefits.
Why Is OIRA Increasingly Important?
Since Reagan first equipped OIRA with the authority to review regulatory actions, the office has been used as a vehicle for advancing and ensuring presidential priorities are in line with the administration’s agenda. For example, when President Trump took office in January 2017, he issued EO 13771, a “2-for-1” regulatory policy that set a regulatory budget and required executive agencies to remove two existing regulations for every regulation they introduced. Tasked with overseeing the implementation and enforcement of these requirements, OIRA had tremendous power in helping the Trump Administration to carry out its deregulatory priorities in the name of business. Today, OIRA plays that same role in guaranteeing the outcome of policies important to the Biden Administration – energy and environment, health care and labor, to name a few.
Although Biden revoked Trump’s EO, he has expanded OIRA’s responsibilities. In his Modernizing Regulatory Review memorandum, Biden directed OIRA to take on “a more proactive role” and work with agencies to craft recommendations on how the regulatory review process can better serve and promote the public interest. This marked an important shift for OIRA. Traditionally, OIRA has held a reactive role in the regulatory review process as opposed to collaborating with executive agencies to formulate regulatory actions from the start.
However, there are potential caveats to OIRA’s regulatory authority. The Supreme Court’s recent decision to corral the EPA’s authority under the Clean Air Act in West Virginia v. EPA based on the major questions doctrine has many wondering whether other agencies could soon face restrictions in establishing new regulations when legislative text or congressional intent produces ambiguity. What would OIRA’s regulatory oversight truly be if agencies were barred from proposing certain regulations? Would OIRA’s ability to coordinate regulatory actions with presidential priorities suffer? These are questions that the administration must contend with as election season approaches and OIRA’s authority potentially becomes even more significant.
While much remains to be seen, one thing is clear: OIRA could be under the brightest stadium spotlight in years as the Biden Administration faces the prospect of delivering its campaign objectives via regulation. Yet, as businesses and non-profits wrestle with the onslaught of pending regulations, it’s important to remember that the regulatory process is a long one and just like any good baseball game, sometimes it takes extra innings to win the game.
